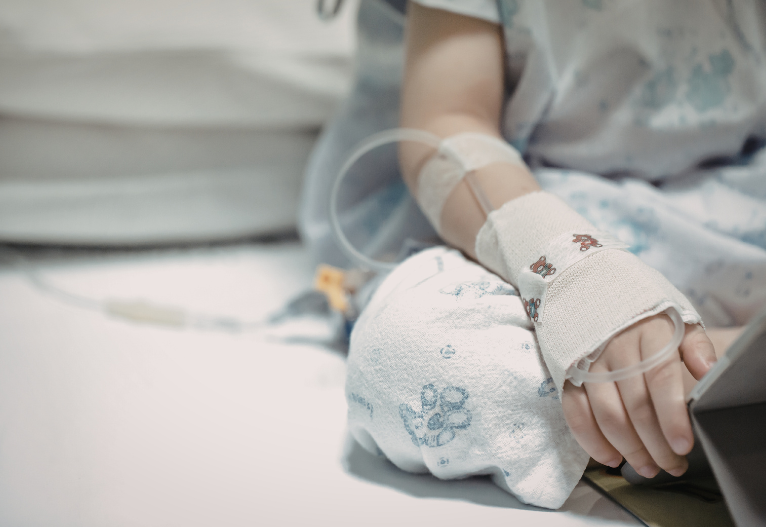
WARNING to parents to stop using homeopathic teething tablets and gels as they pose serious risks to infants.
We originally shared in October 2016, that the U.S. Food and Drug Administration is warning parents to stop using homeopathic teething tablets and gels immediately and throw them away.
May 2018
The Food and Drug Administration has this week said that various gels and creams containing the drug benzocaine can cause rare but deadly side effects in children, especially those two years and younger.
Teething products with benzocaine include Baby Orajel. The packaging states: ‘Instant relief for teething pain.’ The Australian equivalent is Bonjela.
The agency has been warning about the products for a decade but said reports of illnesses and deaths have continued, reports Daily Mail.
Now, it wants teething products off the market, noting there is little evidence they actually work.
‘We urge parents, caregivers and retailers who sell them to heed our warnings and not use over-the-counter products containing benzocaine for teething pain,’ said FDA Commissioner Scott Gottlieb, in a statement.
The FDA said it will take legal action against companies that don’t voluntarily remove their products for young children. Manufacturers are expected to comply as soon as possible.
Please note: I don’t believe the Australian product does include benzocaine ? Correct me if I am wrong.
Previously reported…
“Consumers should seek medical care immediately if their child experiences seizures, difficulty breathing, lethargy, excessive sleepiness, muscle weakness, skin flushing, constipation, difficulty urinating, or agitation,” the government agency wrote in a press release this week. The teething tablets and gels are used on infants and toddlers and claim to help kids deal with the often painful process of teething. They’re incredibly easy to find and are distributed by CVS, Hyland’s and others.
Despite being able to find them almost anywhere, the products haven’t been evaluated or approved by the FDA. Health officials at the agency explained that they can’t say the homeopathic teething tablets and gels are safe or effective. While the makers of these popular teething products claim they help, the FDA told reporters that “the agency is also not aware of any proven health benefit of the products.”
In 2010, the FDA warned parents not to use homeopathic teething tablets and gels after it allegedly received reports of babies having experienced negative side effects – including seizures – from the products. While the FDA is still testing product samples it did release a safety alert that said “Hyland’s Teething Tablets contained inconsistent amounts of belladonna, a substance that can cause serious harm at larger doses,” CBS News reported.
We also UPDATED on 20 Feb 2017
The US Food and Drug Administration issued another warning last month that Hyland’s homeopathic teething tablets contained inconsistent amounts of the “toxic” substance belladonna and posed a risk to children.
The Age shared concerns that numerous pharmacies stock its teething products in Australia, including Terry White Chemists and Priceline.
Fairfax Media actually found Hyland’s teething products for sale at Drew Wood Pharmacy at the Royal Children’s Hospital on Thursday.
“The body’s response to belladonna in children under two years of age is unpredictable and puts them at unnecessary risk,” FDA Center for Drug Evaluation and Research director Janet Woodcock said.
“We recommend that parents and caregivers not give these homeopathic teething tablets to children and seek advice from their health care professional for safe alternatives.”
Australian Skeptics member Peter Hogan, who has been investigating the sale of Hyland’s teething products in Australia, said he was concerned TGA testing was inadequate.
“I was quite shocked that the [pharmacy at the] Royal Children’s Hospital was selling the stuff,” he said.
“I’m concerned parents may be gambling with their babies’ health.”
“Consumers are encouraged to be cautious in purchasing homoeopathic teething products and to talk to their health professional before using them for infants or children.”
Belladonna, also known as deadly nightshade, is a plant that has been used in cosmetics and herbal medicine since the middle ages.
It is also used as a recreational drug because of the hallucinations it can produce.
If parents do want to help their children seek relief from the pain, it is recommended speaking to their doctor about safe alternatives, such as low doses of acetaminophen and ibuprofen.
“That’s normally very helpful around bedtime or nap time to help with the pain,” he said. “But you’d probably want to talk to their pediatrician to make sure you get the dose right.”
You can also freeze everything from teethers, to wet washcloths, to carrots and celery. All of them can numb tiny little aching gums.
UPDATE 1 May 2017
RECALL issued
A big recall is underway, including Australia, after the teething tablets were linked to 10 baby deaths in America.
The Therapeutic Goods Administration (TGA) wants all Hyland’s homeopathic teething and baby nighttime teething tabs to be returned to their place of purchase, because of fears about the effects of the potentially toxic plant belladonna.
TGA published a notice in their bulletin on Saturday, which including this:
“While the TGA had tested samples of these products supplied in Australia and found no quality issues, Kadac is now recalling the tablets as a precautionary measure due to the potential safety risk that belladonna alkaloids can pose to children.”
“The effects of belladonna can be unpredictable and could cause serious health problems.”
Information for consumers
If you have any Hyland’s Baby teething tablets or Hyland’s Baby nighttime teething tablets, do not use them. Return them to the place of purchase for a refund or call Kadac on 1300 762 025.
If you have any questions or concerns about this issue, talk to your health professional.
Share your comments below.
Read more:



















11:13 am
8:36 am
2:32 pm
11:10 am
10:17 am
8:44 am
6:49 am
8:52 pm
8:24 pm
3:52 pm
5:47 pm
2:27 pm
-

-
-
mom81879 replied
- 05 Jun 2018 , 8:37 am
Reply1:23 pm
8:48 pm
7:43 pm
2:33 pm
12:58 pm
12:57 pm
1:30 am
4:27 pm
- 1
- 2
- »
Post a commentTo post a review/comment please join us or login so we can allocate your points.